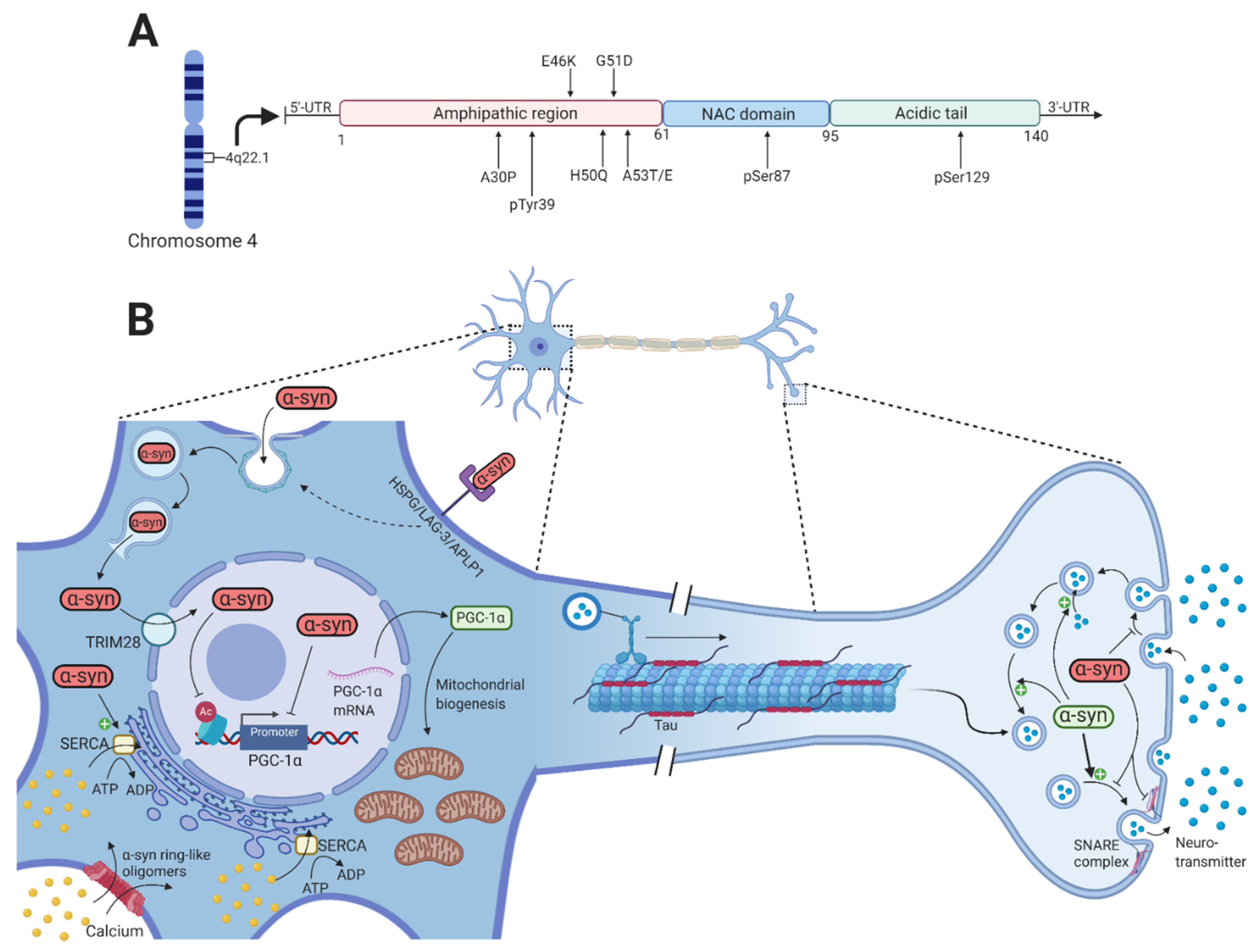
Cells | Free Full-Text | Interaction between Parkin and α-Synuclein in PARK2-Mediated Parkinson's Disease
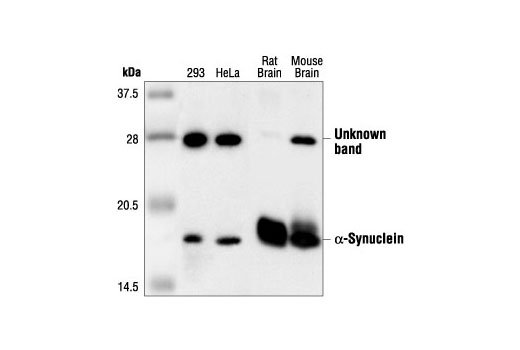
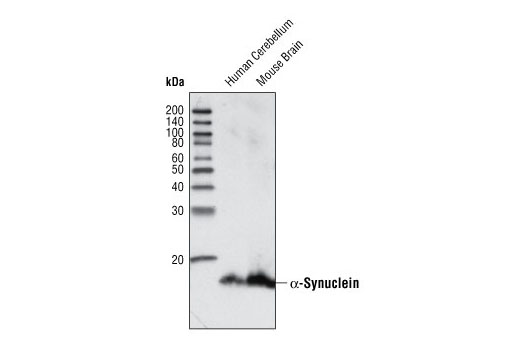
![alpha-Synuclein [Nitrate Tyr125, Nitrate Tyr133] Antibody (24.8) - Azide and BSA Free (NBP2-80572): Novus Biologicals alpha-Synuclein [Nitrate Tyr125, Nitrate Tyr133] Antibody (24.8) - Azide and BSA Free (NBP2-80572): Novus Biologicals](https://images.novusbio.com/fullsize2/alpha-Synuclein-Antibody-(24.8)---Azide-and-BSA-Free-Western-Blot-NBP2-80572-img0001.jpg)
alpha-Synuclein [Nitrate Tyr125, Nitrate Tyr133] Antibody (24.8) - Azide and BSA Free (NBP2-80572): Novus Biologicals

Sequence annotations for alpha-synuclein (UniProt entry: P37840). (a)... | Download Scientific Diagram

Elevated concentrations cause upright alpha-synuclein conformation at lipid interfaces | Nature Communications
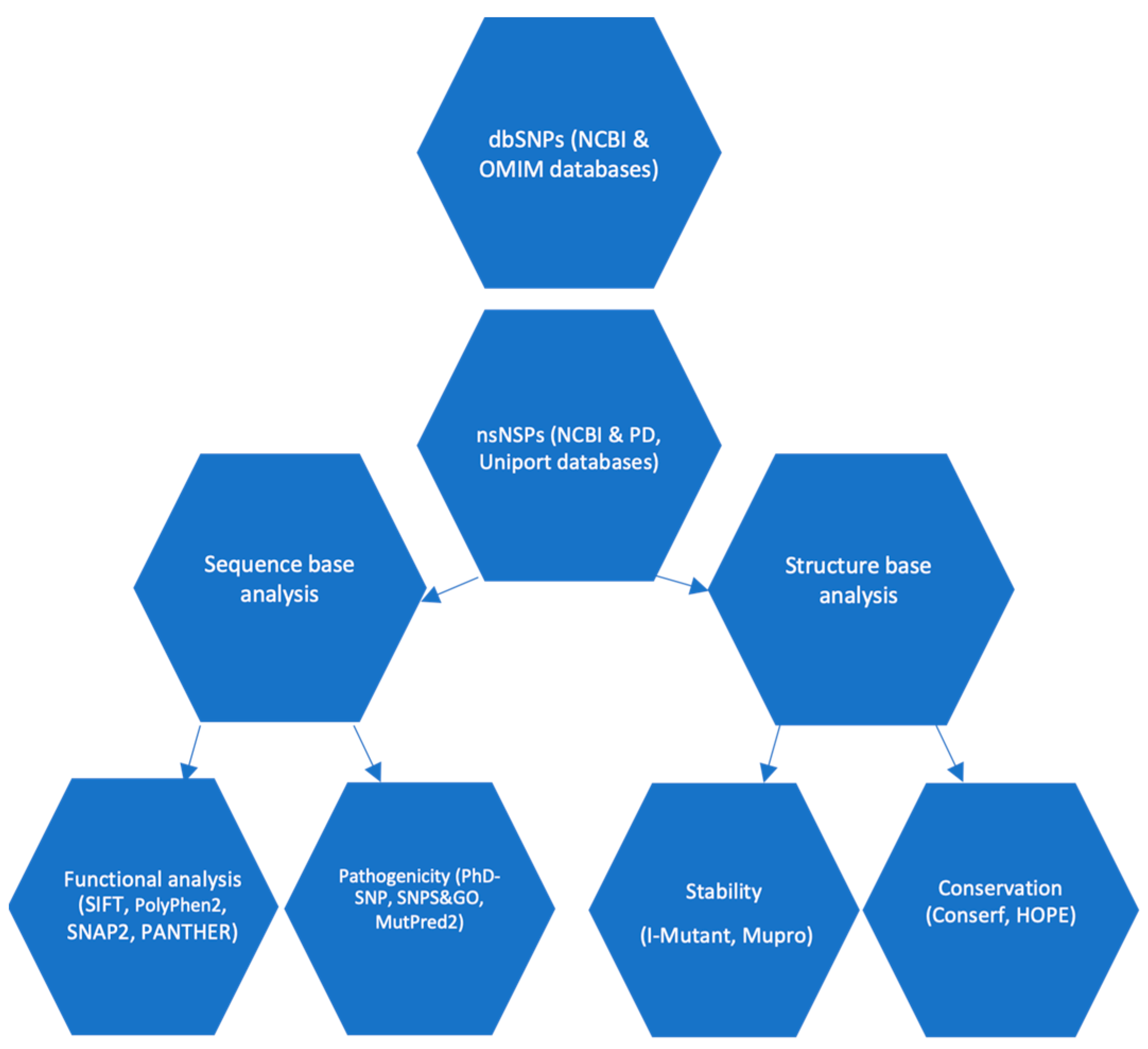
Diseases | Free Full-Text | In Silico Evaluation of the Potential Association of the Pathogenic Mutations of Alpha Synuclein Protein with Induction of Synucleinopathies
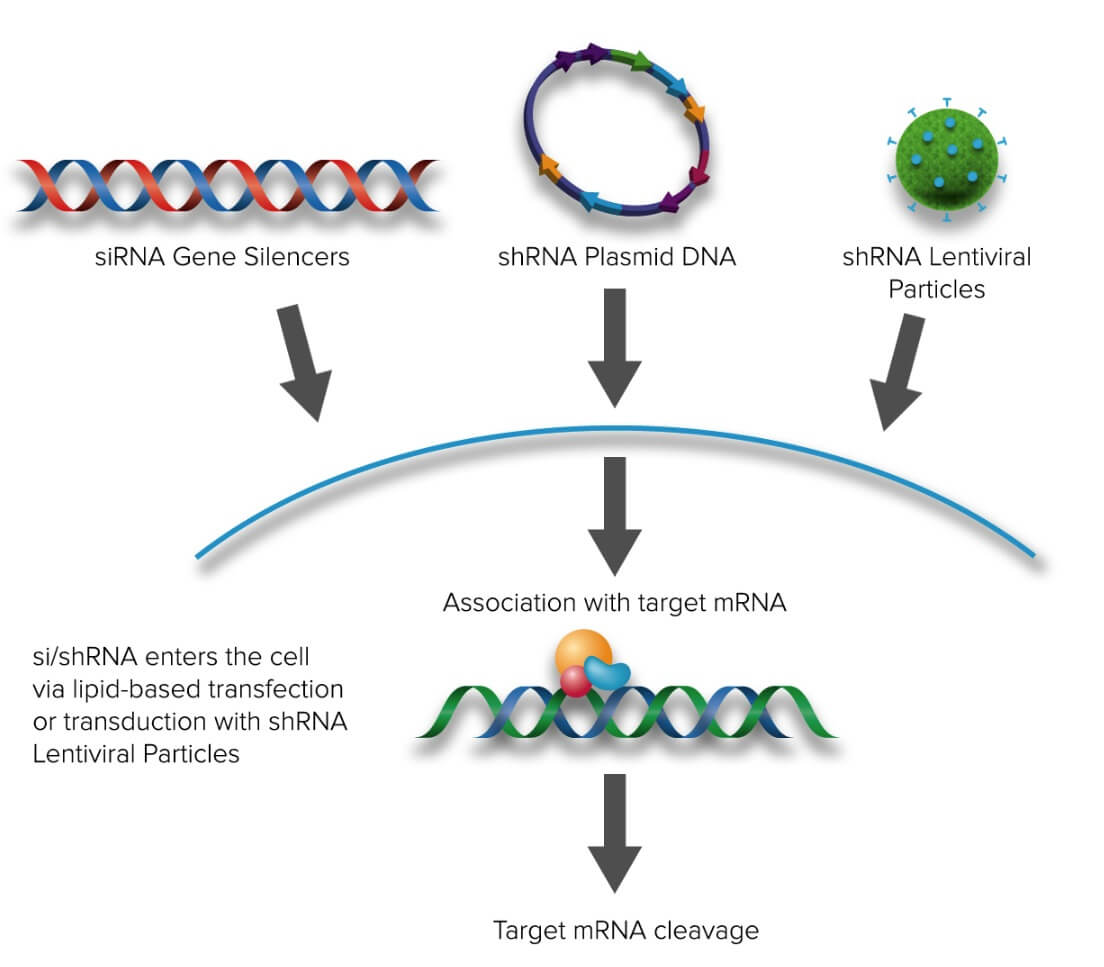
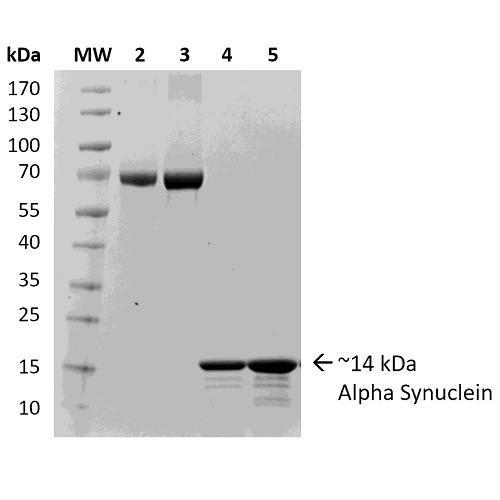
alpha Synuclein siRNA (h), shRNA and Lentiviral Particle Gene Silencers | SCBT - Santa Cruz Biotechnology

The Parkinson's disease protein alpha-synuclein is a modulator of processing bodies and mRNA stability - ScienceDirect
![Recombinant Anti-Alpha-synuclein aggregate antibody [MJFR-14-6-4-2] - Conformation-Specific (ab209538) Recombinant Anti-Alpha-synuclein aggregate antibody [MJFR-14-6-4-2] - Conformation-Specific (ab209538)](https://www.abcam.com/ps/products/209/ab209538/Images/ab209538-459024-ab21403311antialphasynucleinaggregateantibodymjfr14642conformationspecificalexafluor647immunohistochemistryparkinsonsubstantianigrahuman.jpg)
Recombinant Anti-Alpha-synuclein aggregate antibody [MJFR-14-6-4-2] - Conformation-Specific (ab209538)
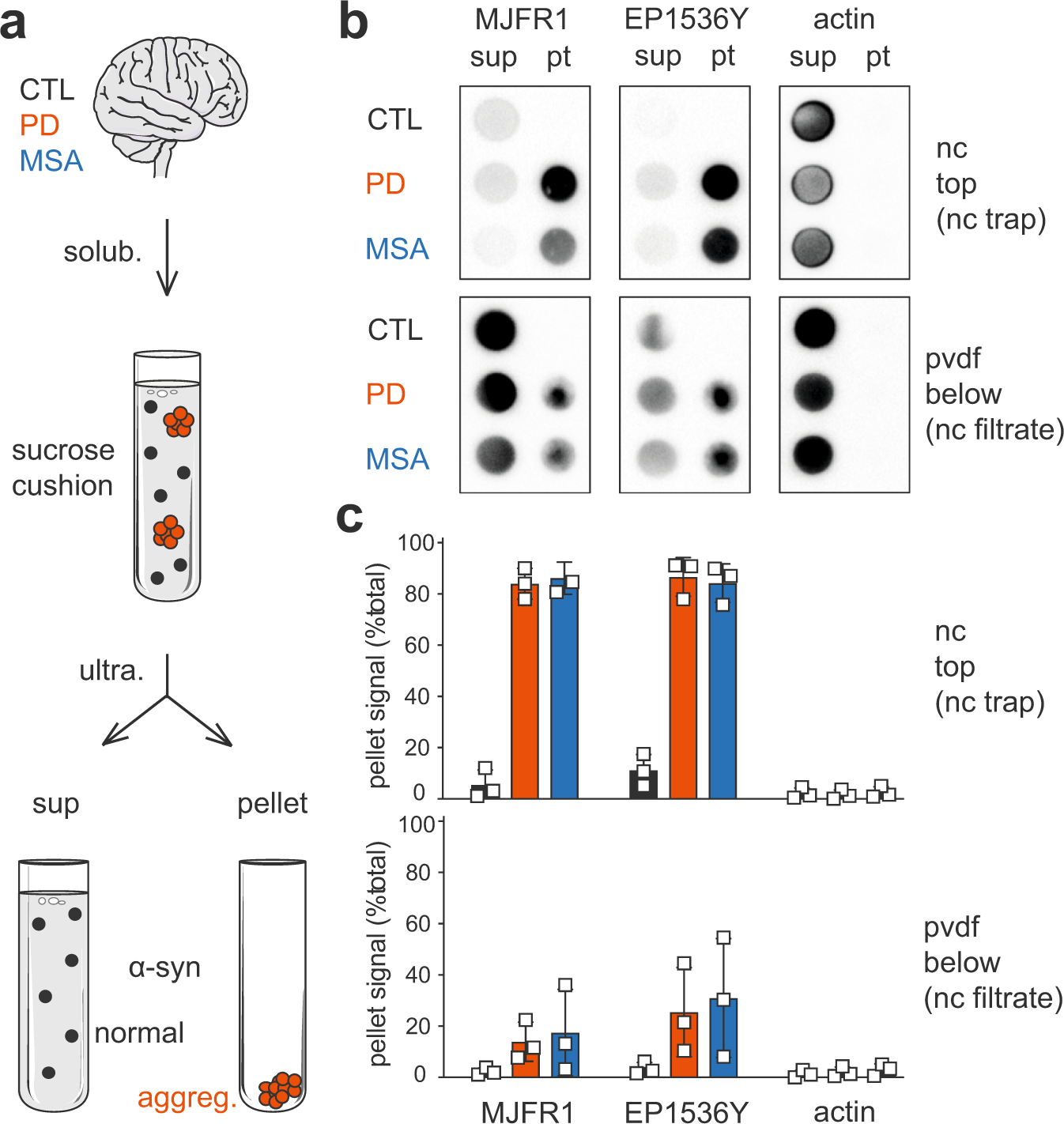
Similar neuronal imprint and no cross-seeded fibrils in α-synuclein aggregates from MSA and Parkinson's disease | npj Parkinson's Disease
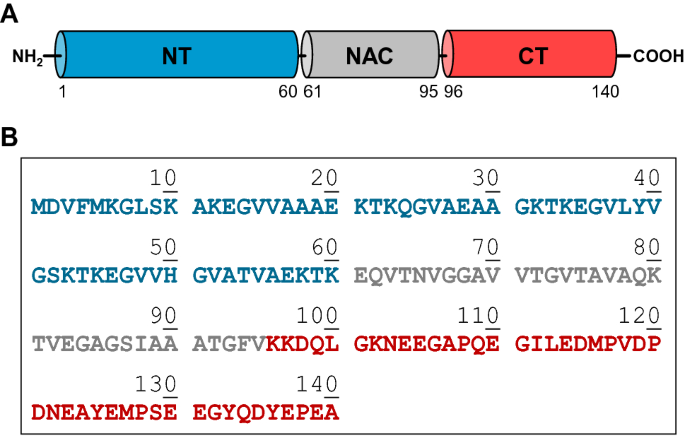
Exogenous human α-Synuclein acts in vitro as a mild platelet antiaggregant inhibiting α-thrombin-induced platelet activation | Scientific Reports

The blobulator: a webtool for identification and visual exploration of hydrophobic modularity in protein sequences | bioRxiv
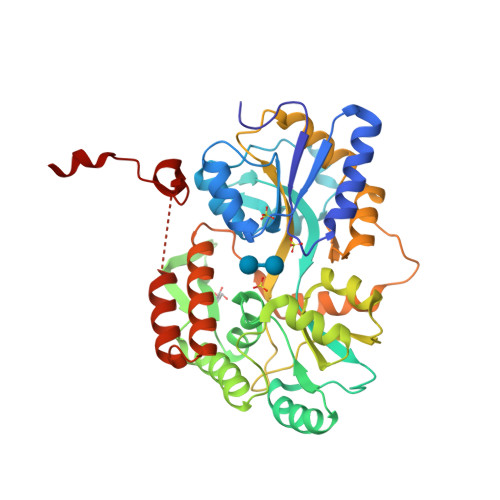
RCSB PDB - 3Q27: Cyrstal structure of human alpha-synuclein (32-57) fused to maltose binding protein (MBP)





![alpha-Synuclein [Nitrate Tyr125, Nitrate Tyr133] Antibody (24.8) (NBP1-26380): Novus Biologicals alpha-Synuclein [Nitrate Tyr125, Nitrate Tyr133] Antibody (24.8) (NBP1-26380): Novus Biologicals](https://images.novusbio.com/images/alpha-Synuclein-Nitrate-Tyr125-Nitrate-Tyr133-Antibody-24-8-Immunohistochemistry-Paraffin-NBP1-26380-img0012.jpg)